When using the ultramicrotome for sectioning, remember to turn on the compressed nitrogen to the air table.
- Following trapezoid trim, place the Epon block, which is still in the chuck, into the specimen arm of ultramicrotome and secure tightly.
- Set the block/specimen arm angle to 0 degrees.
- Place a glass knife into the knife holder and secure tightly. This glass knife will be used to “rough face” the block. Rough facing means removing most, if not all the Epon lying over the tissue in an Epon block in um increments on the ultramicrotome. A glass knife is used for this job because it is inexpensive and replaceable.
- Set the knife clearance angle between 4 to 6 degrees.
- Set the knife stage angle to 0 degrees.
- Set the cutting speed to no more than 1 mm/sec on the Ultracut T. For the Ultracut E, a faster speed can be used (up to 3 mm/sec).
- Set the section thickness to no more than 1 micron (um) on the Ultracut T. The Ultracut E can be more accommodating and can handle up to 3-um increments.
- Advance the left edge of the glass knife to within several millimeters (mm) of the block face. The left edge of a glass knife is the straighter and better edge of the knife to use in thick- and thin sectioning. Look through the stereomicroscope/binoculars to judge this distance approximately. Then lock the stage securely.
- Set the length of the cutting window. To do this on the Ultracut T, note the “window” area of the control panel. There is an upper and lower button.
- Hand rotate the hand wheel clockwise to position the block several mm above the level of the knife-edge. Then depress the upper button once.
- Position the block several mm below the knife-edge by rotating the hand wheel clockwise. Then depress the lower button once. NEVER ROTATE THIS HAND WHEEL COUNTERCLOCKWISE, UNLESS IT IS FOR A FEW MILLIMETERS ONLY!!! THIS HAND WHEEL CAN ACCOMMODATE A FEW MILLIMETERS COUNTERCLOCKWISE TURN.
- To check whether this is an acceptable length of the cutting stroke, turn on the motor to the ultramicrotome and observe the cutting motion. The cutting stroke should commence when the block is several mm above the knife-edge. The end of the cutting stroke is denoted when the entire block face is several mm below the knife-edge. Repeat steps #9 through #12 for the Ultracut T if re-adjustments are needed.
-
To set the length of the cutting window on the Ultracut E, firstly, lock the hand wheel in place. Once the hand wheel is in the locked mode, turn the hand wheel a bit clockwise or counterclockwise to bring the block below the level of the knife. Then set the desired length of the cutting window in mm by adjusting the lever on the hand wheel. Unlock the hand wheel. Turn on the microtome and observe the cutting motion. If the length of the cutting window is longer than needed or too short, then turn off the microtome. Change the cutting length by locking the hand wheel again, then adjusting the lever on the hand wheel to a different value in mm. Turn the microtome on again and assess the new cutting length. When finished, turn off the microtome.
TWO IMPORTANT RULES IN MICROTOMY:
1) ALWAYS RETRACT THE KNIFE AWAY FROM THE BLOCK FACE WHEN MAKING ANY ADJUSTMENTS TO AVOID ACCIDENTAL COLLISIONS.
2) PROPERLY TIGHTEN AND SECURE ALL COMPONENTS OF BLOCK HOLDER, KNIFE HOLDER AND STAGE HOLDER TO ENSURE SAFE AND PROPER USAGE OF THE ULTRAMICROTOME.
- The next step is to align the block face with the knife-edge (left edge of the glass knife). For now, the block sits in a 0-degree angle, as well as the knife stage. We will assume, for now, that the tissue is oriented perfectly perpendicular to the knife. Turn off the fluorescent overhead light in the microtome and use the bottom light source only. Advance the knife-edge close to the block face. Increase the magnification up to 4X. The knife-edge, when brought into close proximity with the block face, will cast a reflection onto the block face. The closer the proximity, the narrower the white shadow becomes. This occurrence is what makes an alignment possible. Regardless of whether the block face is uneven, due to razor trimming or smooth and even, there will be some degree of a white shadow seen. We use this shadow to judge how distant or close the areas of the block face are to the knife-edge. If block and knife-edge are in perfect alignment in terms of the knife stage angle, then the white shadow appears as a thin, even, horizontal white line of light (horizontal alignment). See diagram of horizontal alignment in step #21. If the block face is in perfect vertical alignment, then as the block is put through the cutting motion (manually rotate the hand wheel clockwise), the white shadow reflection continues to appear as a thin white line. If the vertical alignment was off, this means the top or bottom of the block is to close or too far from the knife-edge. Then the block angle must be adjusted to a setting other than 0 degrees.
- As stated, we will proceed with the block/stage being in the 0,0 degree setting. Therefore, we will “face” or trim the block face as it is. To do this, bring the knife-edge as close as possible to the block face without colliding into the block face at any point. Rotate the hand wheel slowly to see where any vertical misalignment lies. If the top or bottom of the block is protruding, then this area will inevitably be cut off first.
- Then, turn on the motor and allow the microtome to remove the overlying Epon in 1 um (or more) increments. Continue to face or trim the block until entire Epon sections are removed. Or stop, once some tissue is seen being cut. Then, retract the knife away from the block face. Use a small vacuum to remove Epon trimmings on and around the microtome.
- The tissue in the block should now be ready for thick sectioning. It may be necessary to re-trim the block to a smaller trapezoid if the block face has gotten too large. Remove the block from specimen arm and with a new razor blade, trim any surrounding Epon, close to the border of the tissue. Or leave a larger margin of Epon around the tissue, depending upon the nature of the experiment. The resulting trapezoid should be no wider that 3 mm at the base and no taller than 3 mm in height. The smaller the trapezoid, the easier it is to section. Return the block to the specimen arm.
- For good thick sections, switch to a new glass knife with a boat attachment or a diamond Histo knife. New students are to continue to use glass knives for all sectioning.
- Place a new glass or a diamond Histo Knife in the holder and secure it tightly. Set the clearance angle between 4 to 6 degrees, generally 6 degrees is most often used. Add enough twice-distilled water to fill the knife boat to make it level with the knife-edge. It is advised to pre-fill the diamond knives with water about ½ hour before use in order to wet down the cutting edge for the sake of convenience. If the diamond knife-edge repels water, then add a few saliva drops to the dry knife-edge for several minutes. This seems to render the diamond knife-edge hydrophilic. Then rinse the knife well under running 2X distilled water. To dry, carefully wick away excess water droplets with filter paper. Avoid touching the cutting edge with anything other than an eyelash hair or fine brush hair. Secure the diamond knife in holder.
- When using a glass knife to thick or thin section, some kind of boat must be attached to the knife. See boat making for glass knives in the protocol entitled, “Glass Knife Making”.
-
Align the new knife to the block face. Look through the binoculars while carefully advancing the knife to vicinity of the block face. It is useful to increase the magnification to up to 4X and use only the bottom light source. Again, note that the knife casts a shadow onto the block face. We will now check the horizontal alignment. The block face surface should now be very even and more reflective. As the knife is advanced even closer, the shadow should appear as a thin white horizontal line across the block face. See left diagram below for a perfect horizontal alignment. The diagram on the right indicates, on the other hand, that knife-edge is too close to the left side of the block surface. Remember to retract knife from block before making any adjustments. Then, to adjust, rotate the knife stage a few degrees to the right. Then, check alignment again. Continue to adjust knife stage as needed.

- Check the vertical alignment. Re-advance the knife to the block face to within a mm or more, while looking through binoculars. Then slowly rotate the hand wheel clockwise to pass the entire height of the block near the knife. Because the block has already been “faced”, the vertical alignment should be fine.
- Now, prepare to thick section the block. Re-check the length of cutting window. Set section thickness setting to 1 um or less. Set the cutting speed to 1 mm/sec. Advance the knife close enough so that the white line shadow becomes a green line. Any closer and the line becomes red. This is close enough. Turn on the microtome and wait for 1 um sections (or less) to begin cutting.
-
The first sections may contain partial tissue and partial Epon. Or the surface tissue may be considered undesirable. Or every section containing tissue is critical. Stop the microtome when enough sections are cut. Remove any unwanted sections from the boat by picking up with a clean, fine artist’s brush. Position the brush under a section(s) and lift out of the water. Save the good sections by transferring them on the brush, one at a time, to a glass microscope slide. Several sections can be placed on a large drop of water on the slide. See diagram of left slide. Or place each section on individual tiny drops of water. As a suggestion, place the deepest cut section apart from the rest, perhaps at the extreme right side on the glass slide. See diagrams:
- Once all the desirable sections have been placed on glass slides, transfer the slides to a hot plate for drying. Set the hot plate temperature hot enough so the sections dry completely in 15 to 20 minutes. If they show too many wrinkles, then they are drying too fast due to too high a temperature setting. If the hot plate is not hot enough, then the sections will not adhere to the slide and will inevitably be rinsed off during staining. To remove unwanted floating sections or debris on the boat water, wipe the surface water with a small piece of Ross Lens Tissue.
- The dried sections should be stained with 1% Toluidine Blue. To stain the sections, apply filtered Toluidine Blue to the sections following their dry down on the hot plate. The stain is 1 part 70% ethanol to 1 part 1% Toluidine Blue. Stain for 30 sec to 2 min, depending on how dark the sections should be. An iridescent rim forms and the stain begins to “smoke” as it begins to evaporate. Remove slide from hot plate and allow it to cool for one minute on the countertop.
- Rinse the sections over a waste container, first with a gentle stream of 2X-distilled water, followed by a stream of 70% ethanol. Lastly, rinse the sections with the water again. Shake off the excess water. Wipe the back of the slide with a Kim wipe, then place the slide back on the hot plate to dry.
- Remove the dried slide from the hot plate and place it on a paper towel on the counter to cool to room temperature. Write in pencil the name of block/tissue, slide #, today’s date, etc., on the label end of slide. Prepare to coverslip it.
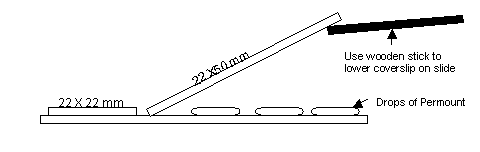
- To coverslip, add 3 to 4 drops of Permount mounting media across the length of the portion of the slide containing the sections. Place one drop on the label end.
-
Slowly lower a glass cover slip (50 mm x 24 mm) at an angle over the section portion of the slide. Avoid trapping any air bubbles in the process. Place a smaller coverslip (22 mm x 22 mm) over the label end. See diagram:
- Place the coverslipped slides in a dust-free area (in a box, e.g.), to dry as is for several days. Then store permanently in a slide box.
- Alternatively, one may wish to examine the slides under a light microscope before coverslipping to examine the stained sections. Studying the section, you may wish to cut deeper into the block for more sections. Or you may not like the present angle of the block if the sections look obliquely cut. Then you must change the block or knife angle or both. If you are keeping track of the length of the tissue then you may wish to remove the block from the microtome and re-measure it. (See Special Procedures at end of this protocol). Whenever a block is removed and returned to the microtome, then it must be re-aligned to the knife as instructed.
- Or return to the block to cut more thick sections and stain them as described.
- When finished with all thick sectioning, the diamond Histo knife must be cleaned before storing away. To do this:
a) First, remove any remaining floating sections in the boat water by wiping the water surface with a Ross lens tissue.
b) Remove water from the boat with a plastic pipette or syringe.
c) Use a clean, razor-beveled end of a Styrofoam stick (usually provided by the manufacturer) that has been lightly soaked in 100% ethanol to do the cleaning as described in the next step.
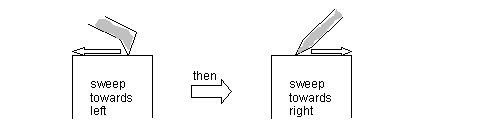
d) Gently and lightly, sweep the beveled Styrofoam end across the length of the diamond knife, starting about 1 mm from the edge, from left to right. Then repeat from right to left. See diagram:
e) Bring the knife over to the 2X-distilled faucet and rinse the knife under 2X distilled running water.
f) Examine the knife-edge under the stereomicroscope of the microtome for any residual debris.
g) If any, then re-clean as directed. See these same instructions from the diamond knife manufacturer’s manual, Diatome, US, e.g.
h) If completely finished, then store the diamond knife in its box.
i) Remember to turn off the compressed N2 to the air table!
SPECIAL PROCEDURES
Tissue measurement in Epon flat blocks
- Place the untrimmed or trimmed flat block, tissue side up, on a glass slide under the light microscope (Nikon Labophot-2).
- Turn on the microscope’s light source and focus on the tissue under 10X objective. Position the block so that the length of tissue can be measured with the eyepiece reticule (focus this eyepiece reticule).
-
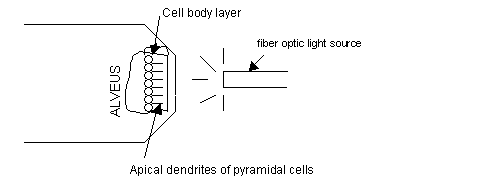
To enhance illumination of the tissue, shine the fiber optic light source near it. One can see right through the hippocampal CA1 pyramidal cell body layer in a 100-micron thick or less tissue slice. This reference is advantageous in measuring distance from the cell body layer. See diagram:
- The magnification values under the objective lenses are as follows: Plan 2: each small division is equal to 50 microns
Plan 10: each small division is equal to 10 microns
Plan 20: each small division is equal to 5 microns
Plan 40: each small division is equal to 2.5 microns - The Epon block can continued to be measured in this way after thick and thin sectioning. In addition, the block can be photographed. See protocol on “Light Microscopy Photography”.